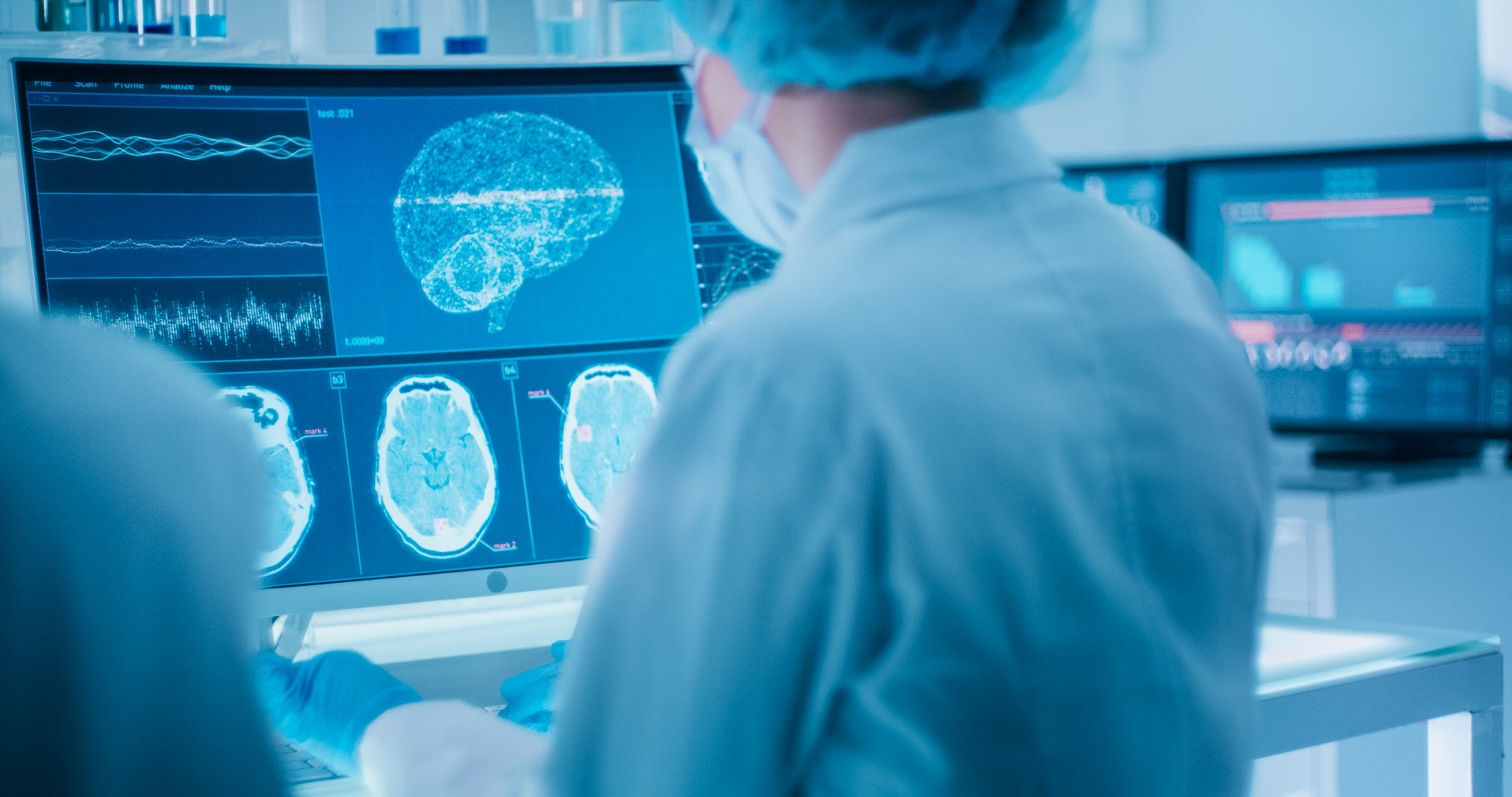
Precision Neuroscience, a company developing brain-computer interface (BCI) technology, has completed the first-in-human pilot of its neural implant system.
As part of the study, surgeons at West Virginia University’s Rockefeller Neuroscience Institute (WVU RNI) temporarily placed Precision’s device on the brains of three patients.

Access deeper industry intelligence
Experience unmatched clarity with a single platform that combines unique data, AI, and human expertise.
In the first phase, the study will include five patients.
Called the Layer 7 Cortical Interface, the device is designed to read, record and map the electrical activity from the brain surface.
The device has 1,024 minuscule electrodes, covering an area of one centimetre square, embedded in a flexible film that conforms to the surface of the brain.
It is designed to be implanted and withdrawn by neurosurgeons without harming brain tissue and is one-fifth the thickness of a human hair.
Even though it was only a small pilot, it advances Precision’s efforts to develop a BCI, a technology that allows brain signals to control external devices.
Precision Neuroscience chief science officer and neurosurgeon Benjamin Rapoport said: “Precision technology has the potential to redefine the standard of care in clinical neuroscience. In the years ahead, we hope to restore function to people with a number of neurological conditions.”
RNI Department of Neurosurgery chairman Peter Konrad said: “This is a remarkable achievement in real-time detection of electrical brain activity mapped with such high resolution. It is as if I was seeing the patient’s brain think.”
In addition, Konrad is the lead researcher overseeing the study at West Virginia University Medicine in partnership with Precision Neuroscience.
The company did not need the Food and Drug Administration (FDA) approval to carry out the trial because the risks were low.
Precision intends to file a request for FDA clearance of their product in the next months, enabling its usage in diagnostic electrophysiologic mapping procedures lasting up to 30 days.
Recently, Elong Musk’s BCI company Neuralink received FDA clearance to proceed with first-in-human clinical trials.







